|
Intravenous injection], and dactinomycin 1♵ mg/m 2 on day 1 given as a single intravenous injection) or four cycles of IVA with doxorubicinģ0 mg/m 2 given as a 4-h intravenous infusion on days 1 and 2 followed by five cycles of IVA. Were randomly assigned (1:1) to receive either nine cycles of IVA (ifosfamide 3 g/m 2 given as a 3-h intravenous infusion on days 1 and 2, vincristine 1♵ mg/m 2 weekly during the first 7 weeks then only on day 1 of each cycle [given as a single Nodal involvement were considered at high risk of relapse. 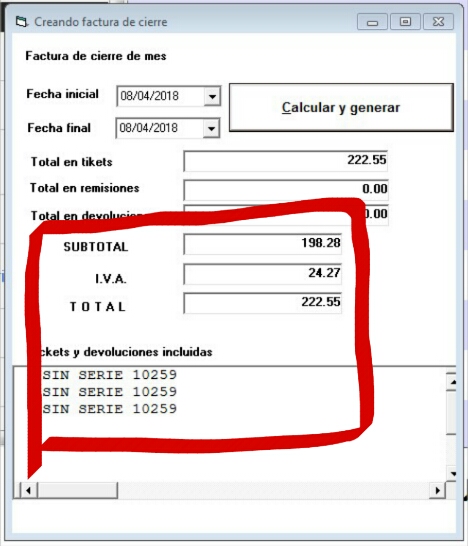
Sites with or without nodal involvement, or those with alveolar rhabdomyosarcoma without Those with embryonal rhabdomyosarcoma incompletely resected and localised at unfavourable We assignedĮach patient to a specific subgroup according to the EpSSG stratification system. 
Than 21 years with a pathologically proven diagnosis of rhabdomyosarcoma. We included patients older than 6 months but younger 
We did a multicentre, open-label, randomised controlled, phase 3 trial involving 108 The Lancet Regional Health – Western Pacific. 
The Lancet Regional Health – Southeast Asia.The Lancet Gastroenterology & Hepatology.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
